A Multiple Ascending Dose Trial Investigating Safety Tolerability And Pharmacokinetics Of Nnc0361
open to eligible people ages 18-45
The trial is a placebo-controlled, double-blinded within cohorts, randomized, multiple ascending dose trial with a sequential trial design. The primary outcome is to investigate the safety and tolerability of ascending subcutaneous weekly doses of NNC0361-0041 plasmid in patients with T1D.
San Francisco, California
Advancing Understanding Of Transportation Options
Sorry, in progress, not accepting new patients
This Stage II randomized, controlled, longitudinal trial seeks to assess the acceptability, feasibility, and effects of a driving decision aid use among geriatric patients and providers. This multi-site trial will test the driving decision aid in improving decision making and quality and determine its effects on specific subpopulations of older drivers . The overarching hypotheses are that the DDA will help older adults make high-quality decisions, which will mitigate the negative psychosocial impacts of driving reduction, and that optimal DDA use will target certain populations and settings.
La Jolla, California
Can Vitamin D Reverse Type 1 Diabetes
A 2010 study found that treatment with vitamin D led to improved glycemic control and insulin sensitivity in people with type 1 and type 2 diabetes. Specifically, increasing vitamin D levels from 25 to 75 nmol/L resulted in a 60% improvement in insulin sensitivity. You can find the most recent type 1 diabetes clinical trials by using Power, which allows you to search trials tailored to your condition, location, and ideal treatment.
Also Check: Do I Need Prescription For Insulin
Join The All Of Us Research Program
JDRF is proud to partner with the National Institutes of the Health on All of Us Research Program. The goal of this program is to gather health information from more than a million Americans to accelerate precision medicine research and medical breakthroughs, and improve healthand it’s critical that the T1D community is represented.
Does Type 1 Diabetes Get Worse With Age
A 2014 study found that those with an earlier onset of type 1 diabetes are associated with a longer burden of disease and more complications as they age. Complications are related to worse glycemic control, renal disease, and severe hypoglycemia, all of which should be considered when creating management plans for older adults with type 1 diabetes. If you are looking to join a type 1 diabetes clinical trial, Power can help you search recent trials by condition, treatment, or location.
You May Like: Normal Blood Sugar For Type 2 Diabetes
Icipation In The Niddk Repository
Data and samples from DPT-1 are made available to researchers through the NIDDK Repository . DPT-1 data, DNA samples, including whole genome amplified samples, and residual serum samples from OGTT tests from enrolled subjects have been deposited in the NIDDK Repository. Proposals for access to data and samples are accepted at any time, and non-renewable samples are subject to stringent review for scientific merit.
TrialNet NHS samples are being stored at the NIDDK Repository. For example, NHS has samples from over 250 subjects with 5-year T1D risk greater than 50% and from 144 individuals who developed diabetes while being followed in the NHS.
Any investigator can propose an Ancillary Study or a Sub-Study to be conducted either prospectively or using stored samples. Proposals are reviewed by TrialNets Ancillary Studies committee, which considers scientific merit, feasibility, and ability of the proposed study to integrate with TrialNets operation without interfering with study goals. The Ancillary Study Committee has received 18 study proposals and 5 have been approved to date.
Steno Diabetes Dialogue Cards Teaching On Group Education On Blood Sugar Control In Adolescents With Type 1 Diabetes
| The safety and scientific validity of this study is the responsibility of the study sponsor and investigators. Listing a study does not mean it has been evaluated by the U.S. Federal Government. Read our disclaimer for details. |
| First Posted : May 27, 2022Last Update Posted : May 27, 2022 |
- Study Details
Also Check: Physical Symptoms Of Diabetes Type 2
Breath Acetone Level As A Measure Of Diabetes Control
Principal Investigator: Michael Thompson, MD
This research is testing a device that measures the amount of acetone in your breath. Acetone is naturally produced when your body digests fats and carbohydrates. It is thought that there are higher levels in people with diabetes than healthy non-diabetics. This study will look at the levels of breath acetone and compare them to blood sugar levels in people with and without diabetes. You will be compensated for your time.
Why Has The Fda Placed The Clinical Trial On Hold
While the sudden hold on the study may be frustrating, Vertex Pharmaceuticals told Beyond Type 1 that it has not yet received any detailed comments or questions from the FDA.
Were really excited about these recent results, and we see it as an important milestone for the program and the field of diabetes research, explained Felicia Pagliuca, vice president, type 1 diabetes disease area executive at Vertex Pharmaceuticals.
The FDA has told us theyve determined there is insufficient information to support increasing the dosage of VX-880 as the study protocol calls for.
Vertex added that VX-880 evaluations have determined that the overall safety profile of the therapy is positive and it is well-tolerated.
Part A of the study was to evaluate VX-880 at half the targeted dosage before moving to the full dosage, said Altshuler.
An independent safety and monitoring committee reviewed the data from Part A which meant Vertex was clear to move forward with Part B, administering the full dosage to patient 3. Part B was underway when Vertex heard from the FDA.
An independent safety and monitoring committee has reviewed the data from Part A of the trial, and concluded that VX-880 is safe, explained Altshuler. This independent safety review meant Vertex was clear to move forward with Part B, administering the full dosage to patient 3. Part B was underway when Vertex heard from the FDA.
You May Like: Type 1 Diabetes Meal Delivery
Stem Cell Therapy For Type 1 Diabetes: Patient Achieved Insulin Independence On Day 270
Editors Note: The news shared below is from a recent and promising clinical trial. For more information on type 1 diabetes research visit our research portal to learn more about getting involved with clinical trials,.
Read past coverage on Vertex Pharmaceuticals stem cell therapy:
The first patient dosed with VX-880a stem cell-derived potential therapy to treat type 1 diabeteshas achieved groundbreaking insulin independence at day 270 with an HbA1c of 5.2 percent, according to a recent press release.
This means the patient no longer requires exogenous insulin doses and is producing enough endogenous insulin to maintain normal blood sugar levels at day 270 of the trial.
In 2021, Vertex Pharmaceuticals Inc. made headlines with earlier results of the same trial: the same patient had experienced a 91% reduction in their daily need for exogenous insulin at day 90.
Still currently in Phase 1/2 of its clinical trial, there is still one caveat to its success: the patient requires immunosuppression therapy to sustain their insulin production. Vertex, however, is confident it will find better ways to manage the immune system around its proprietary product.
Here, well take a closer look at the latest results, the FDAs instructions to pause the study and the future of immunosuppression for the long-term success of VX-880.
Other Benefits From Trialnet Studies
Characterization of risk:benefit of therapies to alter autoimmunity. While TrialNet enrolls only subjects prior to diagnosis or with residual -cell function, the cure of individuals with longstanding disease through islet transplant or regeneration will require control of autoimmunity regardless of the source of new insulin-secreting cells. Thus, TrialNet studies will inform cell replacement efforts by evaluation of therapies to halt immune mediated beta cell destruction.
Characterization of risk:benefit of tolerogenic strategies in children. Information regarding the impact of TrialNet therapies on the pediatric population will be applicable to clinical investigations in other autoimmune diseases that predominantly affect adults.
Also Check: How Does Diabetes Cause Heart Disease
How Close Is A Cure For Type 1 Diabetes
While there is currently no cure for type 1 diabetes, a cure has long been thought to be possible. Research suggests that type 1 diabetes occurs when someone with a certain combination of genes comes into contact with a certain environmental influence. However, less is known about the changes that occur within these genes and how their function is altered. If you are looking to join a type 1 diabetes clinical trial, Power can help you search recent trials by condition, treatment, or location.
So Far Three Patients Have Been Dosed With Vx

Broken into three parts the clinical trial intends to enroll a total of 17 patients with type 1 diabetes. Part A included two patients receiving ½ the intended full dose of VX-880. In Part B, five patients will receive the full dose of VX-880so far, only one patient has begun Part B. In Part C, the remaining 10 patients will receive the full dose of VX-880.
So far, three patients have received infusions of VX-880.
| Participant | |
| 4 to 5.6% | 260 to 1270 pmol/L |
*C-peptide levels are an indication that your body is producing insulin. In people with type 1 diabetes, C-peptide levels can measure anywhere from undetectable to very low levels.
The length of time between when a patient receives VX-880 and when they begin producing insulin is one of the most exciting and difficult to predict aspects of this trial.
These are the only three patients in history to ever receive VX-880, explained David Altshuler, executive VP, global research and chief scientific officer at Vertex Pharmaceuticals. Were still learning how long it will take for the transplanted cells to adjust and begin producing insulin. It takes time.
With the study suddenly on hold in the U.S., the participants whove yet to receive their first dose of VX-880 will wait until the FDA allows it to resume.
You May Like: Surgical Treatment For Type 2 Diabetes
Diagnosis Criteria For Dm
Although both glucose and HbA1c criteria are available for the diagnosis of diabetes, one of these is usually sufficient to make the diagnosis in the setting of the characteristic clinical picture of polyuria, polydipsia, weight loss, and fatigue. Both an HbA1c test and glucose results consistent with diabetes may be required to make the diagnosis if the clinical picture is not clear.- describes the current diagnostic criteria for diabetes, adapted from the ADA Classification and Diagnosis guidelines, and from Type 1 Diabetes Treatment and guideline.
Type 1 Diabetes Clinical Trial Finder
Use our Type 1 Trial Finder, powered by Antidote, to find clinical trials relevant to you.
The Type 1 Trial Finder tool will take you to the website of Antidote, the digital health company who created and support this tool. They will offer you the choice of entering further details, including an email address.
Recommended Reading: Ways To Stop Diabetes Before It Starts
Behavioral Family Therapy And Type One Diabetes
Sorry, in progress, not accepting new patients
Behavioral family therapy, specifically focused on insuring support for the primary caregiver of a child with type one diabetes mellitus and healthy family dynamics, may improve the child’s glycemic control as measured by hemoglobin A1c level .
at UC Davis
Promising Early Results Show That Longstanding Harvard Stem Cell Institute Research May Have Paved The Way For A Breakthrough Treatment Of Type 1 Diabetes Utilizing Research From The Melton Lab Vertex Pharmaceuticals Has Developed Vx
The patient was treated with a single infusion of VX-880 at half the target dose in conjunction with immunosuppressive therapy. The patient, who was diagnosed with T1D 40 years ago and has been dependent on exogenous insulin, achieved successful engraftment and demonstrated rapid and robust improvements in multiple measures. These included increases in fasting and stimulated C-peptide, improvements in glycemic control, including HbA1c, and decreases in exogenous insulin requirement, signifying the restoration of insulin-producing islet cells.
VX-880 is not only a potential breakthrough in the treatment of T1D, it is also one of the very first demonstrations of the practical application of embryonic stem cells, using stem cells that have been differentiated into functional islets to treat a patient, explained Doug Melton, Ph.D., co-director of HSCI, is the Xander University Professor at Harvard and an Investigator of the Howard Hughes Medical Institute. Unlike prior treatments, this innovative therapy gives the patient functional hormone producing cells that control glucose metabolism. This potentially obviates the lifelong need for patients with diabetes to self-inject insulin as the replacement cells provide the patient with the natural factory to make their own insulin, explained Melton.
Also Check: How Much Is Insulin At Walmart
Clinical Trials And The Type 1 Diabetes Cure
I am often asked the question, Where is the cure for Type 1 diabetes ? For those with long-standing diabetes, we are very close to replicating insulin producing beta cell functionality or the actual replacement of those cells either with closed loop systems with continuous glucose sensor driving an insulin pump, or use of replacement beta cells derived from stem cells.
However, as a Pediatrician, I think the ultimate cure for T1D will be prevention. Why cant we screen and predict who is at risk, and then prevent someone from getting Type 1 diabetes in the first place?
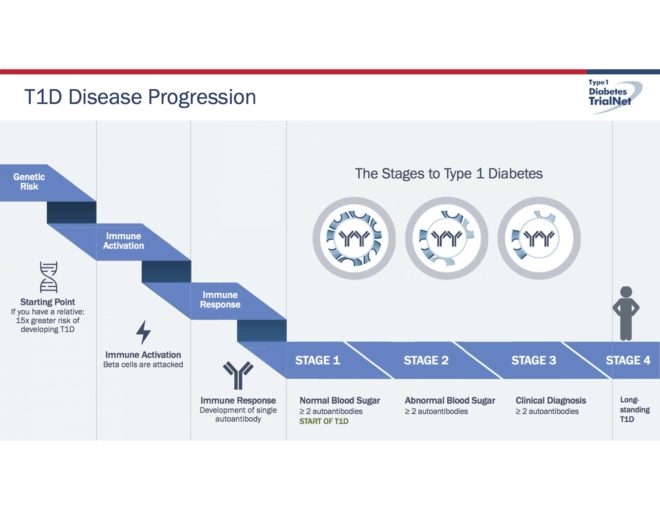
It turns out that T1D occurs in about 1 in 300 people in the general population, but if you already have someone in your family with diabetes, like a brother or sister, then the risk jumps to a 1 in 20 chance of developing Type 1 diabetes. This is why researchers in an NIH sponsored international research effort called TrialNet have been focusing prevention efforts on families with at least one T1D.
We can now use the results from these different tests to determine your overall risk for developing Type 1 diabetes. It turns out that those with two or more autoantibodies will almost assuredly develop T1D eventually. In fact, this is now considered to be the actual start of diabetes, even if the blood sugars are not yet elevated .
To get screened for T1D risk, visit here.
Read Type 1 Diabetes TrialNet.
Metabolic Pathways Of Gra In Patients With Type 1 Diabetes
Sorry, in progress, not accepting new patients
A pilot study for individuals with Type 1 Diabetes who are willing to add a GRA to their current Diabetes treatment regimen. There will be 10 study visits over the course of approximately 8 weeks, with 4 weeks of once weekly, subcutaneous GRA injection. Testing includes 2 MRI scans, 2 glucose challenges, and 2 insulin withdrawal challenges along with physical assessments and vitals.
La Jolla, California
Read Also: Long Term Food Storage For Diabetics
A Safety Tolerability And Efficacy Study Of Vx
| The safety and scientific validity of this study is the responsibility of the study sponsor and investigators. Listing a study does not mean it has been evaluated by the U.S. Federal Government.Know the risks and potential benefits of clinical studies and talk to your health care provider before participating. Read our disclaimer for details. |
| First Posted : March 8, 2021Last Update Posted : March 14, 2022 |
| Diabetes Mellitus, Type 1Impaired Hypoglycemic AwarenessSevere Hypoglycemia | Biological: VX-880 |
| Study Type : | |
| Treatment | |
| Official Title: | A Phase 1/2 Study to Evaluate the Safety, Tolerability, and Efficacy of VX-880 in Subjects Who Have Type 1 Diabetes Mellitus With Impaired Hypoglycemic Awareness and Severe Hypoglycemia |
| Actual Study Start Date : |
| Infused into the hepatic portal vein.Other Name: Formerly known as STx-02 |
Clinical Research In Type 1 Diabetes

Determinants, etiology, progression, prevention, and treatment of Type 1 Diabetes in children and adults.
The Clinical Research in Type 1 Diabetes program includes studies across the lifespan that address the etiology, pathogenesis, prevention and treatment of type 1 diabetes in youth and adults. The program also supports research on hypoglycemia in T1D, including clinical studies and basic research using healthy individuals to understand the physiologic mechanisms of hypoglycemia. The program includes investigator-initiated clinical or behavioral studies, large, multi-center clinical trials that are conducted under cooperative agreements or contracts, and secondary analyses of ongoing clinical trials in diabetes and endocrinology.
Also Check: Treatment Of Type 1 Diabetes In Child
Type 1 Diabetes: Recent Discoveries From Clinical Trials
Diabetes is a disease in which the body does not properly produce insulin, a hormone the body needs to get glucose from the bloodstream into the cells of the body. Glucose is a simple sugar used for energy which is created by the bodyspecifically the pancreasby breaking down the sugars and starches that are consumed.
Type 1 diabetes was previously known as juvenile diabetes. It is usually diagnosed in children and young adults and comprises five percent of all people with diabetes. In Type 1, inflammatory T cells attack beta cells in the pancreas. Beta cells are the insulin producing cells that help keep sugar levels regulated. Many scientists searching for a cure focus on finding ways to prevent the T cell attack on the beta cells.
In one recent type 1 diabetes study, researchers focused on a protein called PD-L1 in blood stem cells that normally limit the T cell attack. Researchers led by those at Harvard Medical Schools Boston Childrens Hospital in Massachusetts discovered that the genetic network of pathways that control PD-L1 production is different in the blood stem cells of diabetics, stopping PD-L1 from being produced. They then experimented with two ways of increasing PD-L1 production, both of which resulted in a diabetes-reversing effect.