Mental Retardation And Autism
In a cohort of 53 children with HNF1B mutations, 3 children were found to have mental retardation and autism, a higher number than expected based on the prevalence in the general pediatric population. Among patients referred for genetic testing for neurodevelopmental or psychiatric disorders, HNF1B mutations were also more frequently present compared with ethnically matched controls.
Diabetes And Exocrine Pancreas Dysfunction
As described above, HNF1B mutations were first described as a cause of MODY. HNF1 has structural similarity to HNF1, the affected gene in the most common type of MODY . Horikawa et al. sequenced the HNF1 transcription factor in 57 patients with MODY and found 1 patient with an HNF1B loss-of-function mutation. HNF1 is thought to play an important role in early development and differentiation of the pancreas. It regulates the expression of key pancreatic proteins, including the HNF4A and SLC2A2 genes, the latter encoding the glucose transporter GLUT2.HNF1B mutations can therefore result not only in pancreatic cell dysfunction, leading to diabetes mellitus, but also pancreatic atrophy and exocrine pancreatic dysfunction. Pancreatic atrophy affecting either the head or body of the pancreas is frequently observed on computed tomography in patients with HNF1B mutations in combination with exocrine pancreatic dysfunction, which is often subclinical. Patients mostly present with diabetes in their early adulthood but with a large variation from the neonatal period to late middle age., Endogenous insulin production is present and patients with diabetes are not obligatorily insulin dependent. However, the majority of patients eventually require insulin therapy for an adequate glycemic control, which is not the case for HNF1-associated MODY. Overall, HNF1B mutations are an infrequent cause of MODY, occurring in < 1% of cases.
Which Individuals Should Be Screened For Hnf1 Mutations
In conclusion, HNF1-related disease is a variable multisystem disorder with limited genotype-phenotype correlation, with specific significance to the nephrologist. It is likely that an unknown but potentially large percentage of HNF1-associated kidney disease is currently not being recognized or is misdiagnosed. In addition, there can be a considerable diagnostic delay before the disease is eventually recognized, as exemplified in some of the presented cases. Although no causal therapy is currently available, diagnosing the syndrome is important. Early recognition makes screening for diabetes, renal function decline, hypomagnesemia, and associated hypokalemia possible, and it may prevent unnecessary examinations and biopsies. In addition, genetic counseling should be offered to patients and their family members.
Also Check: Can You Get Life Insurance With Type 2 Diabetes
Abnormal Liver Function Tests
Elevated liver enzymes are frequent in patients with HNF-1 mutations . This has been characterized by elevation of alanine aminotransferase and -glutamyl transpeptidase without jaundice or liver insufficiency. There have been no liver cysts or other clear aetiological markers seen on ultrasound examination, although in one subject the ultrasound examination suggested liver fibrosis . There have been no reports of liver histology, other than in a 7-month-old child where the histology was normal .
Urine Sample Preparation And Ce

Urine sample collection and CE-MS analysis were performed as reported previously,. Briefly, immediately before preparation, urine samples aliquots stored at 20°C were thawed and 700l were diluted with the same volume of 2M urea, 10mM NH4OH comprising 0.02% SDS. Then, samples were filtered via a Centristat 20-kDa cut-off centrifugal filter device at 2,600g for one hour at 4°C in order to remove high molecular weight compounds. The obtained filtrate was desalted using a PD-10 column equilibrated in 0.01% aqueous NH4OH to eliminate urea, electrolytes and salts. Finally, samples were lyophilized and stored at 4°C prior to CE-MS analysis. The samples were re-suspended in 10µL of HPLC-grade H2O shortly before CE-MS analysis, as described. CE-MS analyses were accomplished using the P/ACE MDQ capillary electrophoresis system online coupled to a MicroTOF MS . The electro-ionization sprayer was grounded, and the ion spray interface potential was defined between 4 and 4.5kV. Spectra were accumulated every 3s along with over a range of m/z to 3503000. Detailed information on accuracy, precision, selectivity, sensitivity, reproducibility and stability of the CE-MS method have been described previously.
Also Check: Patient Teaching On Diabetic Diet
Developmental Abnormalities And Other Rare Features
There are other developmental abnormalities reported in patients with HNF-1 mutations . At present these have usually only occurred in single patients and it is not possible to establish if they are related findings. The high prevalence of developmental anomalies and their distribution in tissues where HNF-1 is expressed , like gut , suggest that, at least for some, the HNF-1 mutation is aetiological. Developmental abnormalities in the lungs have not been described yet in any subjects, but the expression of HNF-1 in lung tissue would suggest that this may occur.
Fig. 1.
The sites of expression and disease manifestations of HNF-1 mutations.
Hyperuricemia And Early Onset Gout
Most patients with HNF1B mutations display hyperuricemia, and some present with early onset gout like our patients 2, 4, 5, and possibly 3. Importantly, for this reason, some patients with HNF1B mutations would meet the diagnostic criteria for familial juvenile hyperuricemic nephropathy. The latter condition is caused by mutations in the UMOD gene, which encodes uromodulin and is suggested to play a role in renal urate transport.HNF1B knockout mice also showed reduced UMOD expression, suggesting that HNF1 regulates transcription of UMOD. Thus, abnormal urate transport could be the reason why patients with HNF1B mutations present with early onset gout and abnormal elevated urate levels discrepant to their level of renal function decline.
Don’t Miss: Best Way To Eat For Diabetes
Molecular Genetics Of Hnf
All reported HNF-1 mutations are shown in . Mutations predominantly cluster in the first five exons. Nonsense, frameshift, splice site and missense mutations have all been described. The intron 2 splice site appears to be a mutational hotspot . Despite heterozygous mutations resulting in the phenotype, not all cases are familial availability of parental DNA has allowed a spontaneous mutation to be established in one case .
Fig. 2.
The distribution of HNF-1 mutations.
There is not a clear correlation of either the type or position of the mutations with the clinical phenotype. The phenotypic variation between family members and between families with the same mutation would point to other genetic or environmental modifiers of the phenotype. A candidate modifier is exposure to maternal hyperglycaemia in utero, which alters age of diagnosis of diabetes in patients with HNF-1 mutations .
Functional Studies On Hnf
Functional studies have been reported on nine HNF-1 mutants. These mutants include one missense mutation, S151P with a change from serine to proline within the DNA-binding region, and two nonsense and five frameshift mutations, all of which are truncating mutations. Seven of these mutants occur within exons 13 and lack all or part of the domains involved in DNA binding and all fail to bind DNA. Two of the mutants in exons 4 and 5 have an intact DNA-binding domain and they bind DNA as efficiently as wild-type protein. The presence of intact DNA binding correlates with the ability to form dimers and transactivate a reporter gene. These mutants have been introduced into Xenopus embryos and all interfere with the development of the pronephros, which is the first kidney in the amphibian. Six of the mutants led to an enlargement of the pronephros. All these mutants lack DNA binding and have no transactivation potential. Three mutants led to a partial or complete agenesis of the pronephros. These included the two mutants with intact DNA binding but also the mutant R137-K161del, which has an in-frame deletion of 24 amino acids within the DNA-binding domain and, thus, fails to bind DNA, although the transactivation domain is intact. The functional studies in Xenopus may define features of the HNF-1 protein which are not detected by in vitro studies .
Don’t Miss: What To Do If You Think Your Diabetic
Poor Renal Function And Uric Acid Levels Were Associated With Srcs In Patients With T2dm Stratified By Age
Since age strongly correlated with SRCs, the association between age and SRCs was further analyzed. The patients were divided into four groups according to age quartiles. According to the ANOVA, the eGFR of patients in the four groups satisfied the homogeneity of variance assumption, and the decrease in the eGFR of the SRC group was more significant than that in the group without SRCs . Regarding proteinuria, except for the group in which the ages ranged from 59 to 67 years old, the incidence of SRCs increased significantly in patients with proteinuria in the different age groups . Patients were divided into four groups according to the quartiles of uric acid levels. As shown in Figure 1D, the prevalence of SRCs significantly increased as the uric acid levels increased . In addition, among patients over 67 years old, the incidence of SRCs was significantly increased in patients with a high level of uric acid compared to patients with normal level of uric acid, but a noticeable difference was not observed in other age groups .
Prenatal Identification Of A Pathogenic In
Prenatal panel sequencing in individual I1 revealed a novel heterozygous 3-bp deletion in exon 3 of HNF1B , resulting in an in-frame deletion of the highly conserved AA glycine at position 239 ) . The identified indel variant was not present in the Genome Aggregation Database and was computationally predicted to be deleterious. A potential splice effect was not detected . The c.715_717del variant could not be identified in DNA from peripheral blood lymphocytes of both parents and thus likely arose de novo. Structural modeling based on the crystal structure of the HNF1B protein showed that the glycine at position 239 lies in an alpha-helix motif of the homeodomain, which has DNA-binding function. The deletion of Gly is predicted to disrupt two neighboring AAs involved in binding to DNA . Two missense variants and c.716G> A, p.) affecting the same AA were reported in a boy with bilateral renal cysts, kidney failure, and diabetes in childhood and in a girl with MODY, renal cysts in the right kidney, agenesis of left kidney, and pancreas atrophy, respectively. Furthermore, a variant affecting the neighboring residue Trp238 ) was described as pathogenic . On the basis of the ACMG criteria , we classified the identified variant as pathogenic .
Figure 2HNF1BHNF1B
Also Check: How Many People In The Us Have Type 1 Diabetes
The Relationship Between Simple Renal Cysts And Renal Function In Patients With Type 2 Diabetes
- Hunan Key Laboratory of Kidney Disease and Blood Purification, Department of Nephrology, The Second Xiangya Hospital, Central South University, Changsha, China
Introduction: Simple renal cysts are the most common acquired cystic kidney disease, but the relationship between SRCs and renal function has not been clarified in patients with type 2 diabetes mellitus .
Methods: A retrospective study was conducted to analyze the clinical features of renal cysts and ultrasound data of the kidney in 4,304 patients with T2DM.
Results: The prevalence of SRCs in patients with T2DM was 21.1%. Compared to patients with no SRCs, patients with SRCs had worse renal function . After adjusting the confounders, SRC was related to estimated glomerular filtration rate in patients with T2DM . Age, gout, proteinuria, cerebrovascular disease , and increased serum phosphorus levels were associated with SRCs in patients with T2DM.
: SRCs are associated with worse renal function in patients with T2DM. More attention should be paid to gout, proteinuria, CVD, serum phosphorus levels, and renal function in T2DM patients with SRCs.
Q12 Microdeletions In Individuals I2 To I7
Postnatal high-resolution CMA in individuals I2 to I7 revealed 17q12 microdeletions of variable sizes ranging from 1.42 Mb in I5 to 1 80 Mb in I6 . De novo occurrence was confirmed in all cases apart from individual I3, where the deletion was excluded in the mother, but paternal DNA was not available .
Don’t Miss: How To Administer Insulin Pen
Renal Cysts And Diabetes Syndrome Autism And Schizophrenia Linked
Researchers have uncovered a small genomic deletion in patients with autism spectrum disorders and schizophrenia.
Researchers whose study results were published yesterday in the American Journal of Human Genetics have uncovered a small genomic deletion in patients with autism spectrum disorders and schizophrenia. The region in which the deletion occurs also includes a gene that when mutated is know to cause renal cysts and diabetes syndrome .
“The genetic overlap between ASD and schizophrenia, both of which have a high heritability, has been the focus of several recent studies however, no single specific genetic cause accounts for more than 1%-2% of cases,” said lead author Dr. Daniel Moreno-De-Luca.
Working with a team of investigators, Moreno-De-Luca analyzed the DNA of 23,000 patients with ASD, developmental delay, or schizophrenia to find DNA duplications or deletions . In 24 patients, the researchers found the same deletion on chromosome 17 , a CNV that was absent in 52,448 controls.
“We calculate the risk for this combined sample to be at least 13.58, and probably much higher,” said Dr. David H. Ledbetter, Emory University.
The gene highlighted in the current study is one of 15 contained within the deletion. Whats interesting is that many patients with ASD in the study had a family history of kidney
disease and/or diabetes. Additionally, patients with RCAD often present with neurodevelopmental disorders.
Related Content:
Citation Doi And Article Data
Citation:DOI:Dr Francis DengRevisions:see full revision historySystems:
- Renal cysts and diabetes syndrome
- HNF1B-associated disease
- Maturity-onset diabetes of the young, type 5
- MODY5
Renal cysts and diabetes syndrome , also known as maturity-onset diabetes of the young, type 5 , refers to the combination of renal cortical cysts and diabetes mellitus in patients with mutations in the HNF1B gene. When renal cysts are associated with these mutations without disturbances in glucose metabolism, the term HNF1B-associated kidney disease may be used.
Read Also: Are Omelettes Good For Diabetics
Heterogenic Presentation Of The Multisystem Phenotype Of Hnf1
The first patient, a woman aged 39 years, was referred to the nephrologist with suspected polycystic kidney disease because of reduced eGFR and renal cysts, without a family history of polycystic kidney disease. Her medical history revealed recurrent urinary tract infections, enuresis nocturna until age 25 years, and type 2 diabetes mellitus and gout at age 32 years. At the time of referral, her serum creatinine was 94 mol/L, uric acid was 0.46 mmol/L, magnesium was 0.79 mmol/L, hemoglobin A1c was 39 mmol/mol while receiving subcutaneous insulin therapy, and endogenous creatinine clearance was approximately 80 ml/min without proteinuria. A renal ultrasound demonstrated multiple cortical as well as parapelvine cysts in both kidneys. The combination of diabetes at a young age and renal cysts was suggestive of HNF1-associated disease. Genetic analysis indeed revealed a previously described c.883C> T p.Arg295Cys HNF1B point mutation in an evolutionary conserved amino acid in the homeobox domain of the HNF1 protein.
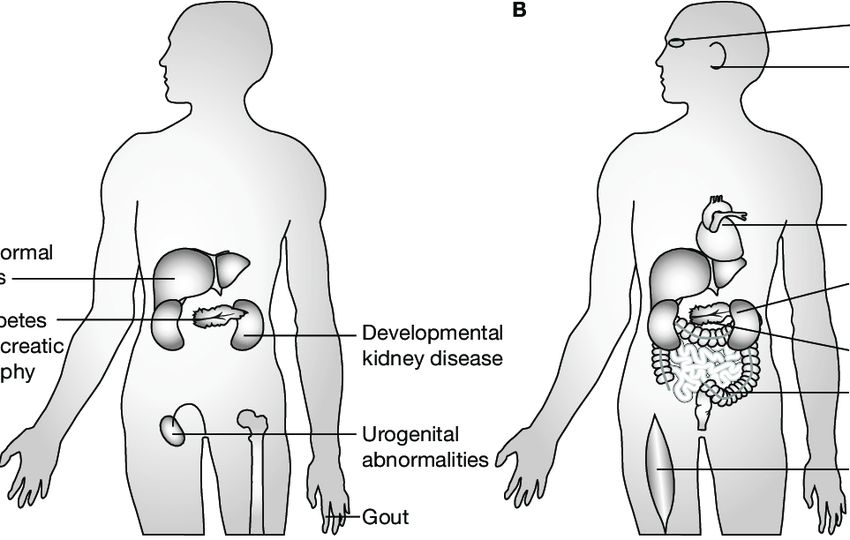
Renal and extrarenal features of HNF1-associated disease. The clinical signs and symptoms that are currently associated with HNF1B mutations and deletions are depicted.
HNF1 as a promiscuous transcription factor. Target genes known to be regulated by the HNF1 transcription factor in several organ systems, responsible for the diverse multisystem clinical signs and symptoms, are depicted.
Srcs Correlated With Age Gout Proteinuria And Cvd In Patients With T2dm
Variables that were significantly different in patients with and without SRCs were included in the logistic analysis , including age, gender, disease duration, waist-hip ratio, hemoglobin, PLT, TG, TC, FPG, HbA1c, SCr, 24 h urinary albumin, calcium, phosphorus, smoking, drinking, urolithiasis, history of gout, DF, DK, hypertension, CVD, fatty liver, and insulin therapy . A multivariate logistic regression analysis was conducted to identify the risk factors for SRCs, and age, gout, proteinuria, CVD, and increased serum phosphorus levels were independent risk factors for SRCs in patients with T2DM. Urolithiasis, postoperative urolithiasis, and normal or decreased phosphorus levels were not significant risk factors for SRCs .
Figure 3. Logistic regression analysis for simple renal cysts. Adjusted confounders variables included gender, age, duration, smoking, drinking, urolithiasis, waist-hip ratio, HB, PLT, TG, Scr, ALB, FPG, HbA1c, 24 h urinary albumin, Ca, P, DK, DF, hypertension, CVD, fatty liver, UTI, CVD, and insulin therapy. SRCs, simple renal cysts SCr, serum creatinine TG, triglyceride CVD, cerebrovascular disease DF, diabetic foot MIAU, microalbuminuria MAAU, microalbuminuria DF, diabetic foot.
Also Check: What Is Worse Diabetes 1 Or Diabetes 2
Disease Mechanisms In Man
In human subjects, the three renal histologies of cystic renal dysplasia, glomerulocystic disease and oligomeganephronia are all the result of abnormal renal development. In human metanephroi, HNF-1 transcripts are predominantly expressed in the collecting ducts with lower levels over the glomeruli . The formation of glomerular cysts might be secondary to another abnormal process occurring during renal development, such as transient microscopic obstruction of immature nephrons. Calyceal abnormalities have been described in HNF-1 mutation carriers with hypoplastic glomerulocystic kidney disease and in a subject with a solitary functioning kidney . Pelviureteric junction obstruction has also been described and one subject required a pyeloplasty . The renal pelvis and calyces are derived from the ureteric bud in the embryo, which is a site of HNF-1 expression .
Renal Cysts And Diabetes Syndrome Rcad
Alternative titles symbols
MATURITY-ONSET DIABETES OF THE YOUNG, TYPE 5 MODY5HYPERURICEMIC NEPHROPATHY, FAMILIAL JUVENILE, ATYPICALFJHN, ATYPICALTUBULOINTERSTITIAL KIDNEY DISEASE, AUTOSOMAL DOMINANT, 3 ADTKD3GLOMERULOCYSTIC KIDNEY DISEASE, HYPOPLASTIC TYPEGLOMERULOCYSTIC KIDNEY, FAMILIAL HYPOPLASTICCONGENITAL ANOMALIES OF THE KIDNEY AND URINARY TRACT WITH DIABETESCAKUT WITH DIABETES
GROWTH ABDOMEN Liver Pancreas GENITOURINARY External Genitalia Internal Genitalia Kidneys SKELETAL ENDOCRINE FEATURES LABORATORY ABNORMALITIES MISCELLANEOUS MOLECULAR BASIS
Tubulointerstitial kidney disease – – 5 Entries
| Location |
|---|
â¼TEXT
A number sign is used with this entry because of evidence that renal cysts and diabetes syndrome is caused by heterozygous mutation in the TCF2 gene on chromosome 17q12.
â¼Description
For a phenotypic description and a discussion of genetic heterogeneity of MODY, see .
For a discussion of genetic heterogeneity of ADTKD and a discussion of the revised nomenclature of these disorders, see ADTKD1 .
The renal abnormalities are part of a spectrum of malformations known as congenital anomalies of the kidney and urinary tract .
â¼Clinical Features
reported a Japanese family in which 3 sibs developed MODY at ages 14, 10, and 15 years, respectively. Their mother and maternal uncle developed diabetes at ages 40 and 60, respectively their father developed diabetes at age 50. A nonspecific nephropathy was described.
Clinical Variability
â¼Molecular Genetics
| 189907 |
Recommended Reading: Does Diabetes Affect Your Stomach