Other Specific Types Of Diabetes
Genetic Defects of the -Cell
Several forms of diabetes are associated with monogenetic defects in -cell function. These forms of diabetes are frequently characterized by onset of hyperglycemia at an early age . They are referred to as maturity-onset diabetes of the young and are characterized by impaired insulin secretion with minimal or no defects in insulin action. They are inherited in an autosomal dominant pattern. Abnormalities at six genetic loci on different chromosomes have been identified to date. The most common form is associated with mutations on chromosome 12 in a hepatic transcription factor referred to as hepatocyte nuclear factor -1. A second form is associated with mutations in the glucokinase gene on chromosome 7p and results in a defective glucokinase molecule. Glucokinase converts glucose to glucose-6-phosphate, the metabolism of which, in turn, stimulates insulin secretion by the -cell. Thus, glucokinase serves as the glucose sensor for the -cell. Because of defects in the glucokinase gene, increased plasma levels of glucose are necessary to elicit normal levels of insulin secretion. The less common forms result from mutations in other transcription factors, including HNF-4, HNF-1, insulin promoter factor -1, and NeuroD1.
Genetic Defects in Insulin Action
Diseases of the Exocrine Pancreas
Endocrinopathies
Drug- or Chemical-Induced Diabetes
Infections
Uncommon Forms of Immune-Mediated Diabetes
B Preclinical And Clinical Studies Examining Pathogenic Relationships Of Diabetes And Nafld
Describing the pathogenesis of NAFLD and NASH in the human with diabetes is complex, and it has been challenging to separate the relative contributions from obesity and other components of the metabolic syndrome. Factors causing NAFLD progression in diabetes are, as yet, not well defined, but they are likely to be an interplay of dysfunctional lipid metabolism and disordered glucose regulation related to insulin resistance with hyperinsulinemia and a relative insulin deficiency, increased oxidative stress, and local and systemic inflammation. Both genetic and environmental conditions are likely to interact to cause NAFLD and NASH and also influence the strong relationship that exists with type 2 diabetes .
1. Genetic factorscommon soil
Intrauterine growth retardation has also been shown to be associated with NAFLD in pediatric populations and may be related to similar epigenetic changes that have been linked to increased type 2 diabetes risk and development of the metabolic syndrome .
2. A seeming paradox: insulin resistance with hyperinsulinemia and a simultaneous relative insulin deficiency in diabetes may cause the progression of NAFLD
a. Disordered lipid metabolism in NAFLD pathogenesis: insulin resistance, diabetes, and hepatic steatosis.
b. Lipid toxicity may progress simple steatosis to NASH.
c. Hepatic inflammation and oxidative stress.
d. Disordered insulin signaling and hepatic fibrosis.
4. Chronic hyperglycemia in diabetes may contribute to NAFLD progression
Ffa And Hepatic Glucose Metabolism
The liver plays a pivotal role in the regulation of glucose metabolism . Following carbohydrate ingestion, the liver suppresses its basal rate of glucose production and takes up approximately one-third of the glucose in the ingested meal .
Collectively, suppression of glucose production and augmentation of hepatic glucose uptake account for the maintenance of nearly one-half of the rise in plasma glucose concentration following ingestion of a carbohydrate meal. Hepatic glucose production is regulated by a number of factors, of which insulin and glucagon and FFA are the most important. In vitro studies have demonstrated that plasma FFA are potent stimulators of endogenous glucose production and do so by increasing the activity of pyruvate carboxylase and phosphoenolpyruvate carboxy-kinase, the rate limiting enzymes for gluconeogenesis . FFA also enhances the activity of glucose-6- phosphatase, the enzyme that ultimately controls the release of glucose by the liver .
Read Also: Can Too Much Thyroid Medication Cause High Blood Sugar
Ethnicity And Family History/genetic Predisposition
Globally, the incidence and prevalence of T2DM are found to vary widely depending on ethnicity and geographical region with Japanese, Hispanics and Native Americans having the highest risks . It has been shown higher incidence rates in Asians compared with a White American population , and white population in the UK, , where the highest risk is among the black population . Whilst no clear reasons have been found, contributing factors such as modern lifestyle factors , socioeconomic and direct genetic propensity or gene environmental interactions have been postulated.
Primary Hypersecretion Of Insulin
An alternative view to explaining the state of insulin resistance is the notion that primary beta cell overstimulation results in insulin hypersecretion. This leads to the development of obesity and insulin resistance, and then, to beta cell exhaustion . In a model that presupposes beta cell hypersecretion as the initial manifestation of beta cell dysfunction, insulin sensitivity is modulated by insulin secretion. When beta cell hypersecretion occurs, the responsiveness of insulin-sensitive tissues to insulin is downregulated and, these tissues become insulin resistant. The latter becomes necessary to maintain normal glucose tolerance, without the adverse outcome of hypoglycemia. However, considering that beta cell hypersecretion is primary and fixed, when insulin sensitivity is acutely improved, hypoglycemia would be expected to ensue. In either case, the demonstration of the existence of a feedback loop that regulates glucose metabolism has made it clear that assessment of the adequacy of beta cell function requires knowledge of both the degree of insulin sensitivity and the magnitude of the insulin response.
Recommended Reading: What Sodas Can Diabetics Drink
Summary: Ffa And The Pathogenesis Of Obesity And T2dm
n obese individuals and in the majority of subjects with T2DM, there is an expanded fat cell mass and the adipocytes are resistant to the anti-lipolytic effects of insulin . Most individuals with obesity or T2DM are characterized by visceral adiposity and visceral fat cells have a high lipolytic rate, which is especially refractory to insulin . Not surprisingly, both T2DM and obesity are characterized by an elevation in the mean day-long plasma FFA concentration. Elevated plasma FFA levels, as well as increased triglyceride/fatty acyl CoA content in muscle, liver, and beta cell, lead to the development of muscle/hepatic insulin resistance and impaired insulin secretion.
Insulin Secretion And Metabolism
The homeostasis of glucose is maintained by a balance of hepatic glucose production and peripheral glucose uptake and utilization. Glucose is the key regulator of insulin secretion by the pancreatic beta cell. Other metabolic end products such as ketones, amino acids, enteric peptides and neurotransmitter also influence insulin release.
Insulin is secreted in a pulsatile pattern with small secretory bursts every 10 minutes superimposed upon greater amplitude oscillations of about 80-150 minutes. After a meal, bursts of insulin secretion usually last for two to three hours prior to returning to baseline levels.
Once insulin is released into the portal venous system, the liver degrades half of the secreted insulin. The remaining insulin enters the systemic circulation to reach the target binding sites. Activation of insulin receptors induces synthesis of glycogen, synthesis of proteins, lipogenesis and regulation of various genes in insulin-responsive cells.
There are three pathophysiologic mechanisms, which are central to the development of Type II diabetes mellitus: impaired insulin secretion, peripheral insulin resistance, and excessive hepatic glucose production. In the early phase of this disease, despite the presence of insulin resistance, blood glucose may remain normal due to compensatory increase in insulin secretion by the remaining beta cells. Once this compensatory mechanism is exhausted, derangements in blood glucose concentrations become evident.
Also Check: Diabetes Feet Tingling At Night
Insulin Receptor Signal Transduction
Following activation, insulin receptor tyrosine kinase phosphorylates specific intracellular proteins, of which at least nine have been identified . Four of these belong to the family of insulin-receptor substrate proteins: IRS-1, IRS-2, IRS-3, IRS-4 . In muscle IRS-1 serves as the major docking protein that interacts with the insulin receptor tyrosine kinase and undergoes tyrosine phosphorylation in regions containing amino acid sequence motifs . When phosphorylated, these serve as recognition sites for proteins containing src-homology 2 domains . Mutation of these specific tyrosines severely impairs the ability of insulin to stimulate glycogen synthesis and DNA synthesis, establishing the important role of IRS-1 in insulin signal transduction . In liver, IRS-2 serves as the primary docking protein that undergoes tyrosine phosphorylation and mediates the effect of insulin on hepatic glucose production, gluconeogenesis and glycogen formation . In adipocytes, Cbl represents another substrate which is phosphorylated following its interaction with the insulin receptor tyrosine kinase, which is required for stimulation of GLUT 4 translocation.
Phosphorylation of Cbl occurs when the CAP/Cbl complex associates with flotillin in caveolae, or lipid rafts, containing insulin receptors .
B Other Agents With Potential Benefit In The Patient With Nash And Diabetes
1. Antioxidants
Despite the positive findings associated with the use of vitamin E, because treatment is likely to be needed long term, some caution must be applied given the results of several meta-analyses suggesting a small but significant increase in risk of all-cause mortality with supplementation, particularly at doses greater than 400 IU/d . Given current evidence, vitamin E has been recommended at a dose of 800 IU/d in patients with active NASH , without diabetes. There is a need for validation of vitamin E as an effective treatment of NASH in patients with diabetes, with close attention paid to long-term safety data .
2. Ursodeoxycholic acid
UDCA has theoretical benefits in NASH through its antiapoptotic properties, by decreasing TNF- levels and perhaps by reducing ER stress and increasing insulin sensitivity. Disappointingly, three placebo-controlled RCTs with UDCA have demonstrated mixed but largely negative results . A subsequent review has suggested the possible benefit of UDCA plus vitamin E and also the examination of non-UDCA bile acids that may regulate metabolism via the farnesoid X receptor , a nuclear receptor involved in sensing bile acids. FXR is thought to be a key regulator of lipid and carbohydrate metabolism .
3. Novel agents
Recommended Reading: What Is Diabetic Autonomic Neuropathy
Iv Nafld And Nash Definitions
NAFLD incorporates a spectrum of pathology from simple steatosis, to nonalcoholic steatohepatitis , to fibrosis and cirrhosis . Patients with NAFLD are also at increased risk of hepatocellular carcinoma . Although NAFLD is likely to have an impact on the prognosis of a variety of other liver diseases , the diagnosis is generally reserved for patients without other significant causes of liver pathology, particularly excluding those with significant alcohol intake.
Summary Of Insulin Resistance In T2dm
Insulin resistance involving both muscle and liver are characteristic features of the glucose intolerance in individuals with T2DM. In the basal state, the liver represents a major site of insulin resistance, and this is reflected by overproduction of glucose despite the presence of both fasting hyperinsulinemia and hyperglycemia. This accelerated rate of hepatic glucose output is the primary determinant of the elevated fasting plasma glucose concentration in T2DM. Although tissue glucose uptake in the post-absorptive state is increased when viewed in absolute terms, the efficiency with which glucose is taken up is diminished. After glucose infusion or ingestion both decreased muscle glucose uptake and impaired suppression of HGP contribute to the insulin resistance. Following glucose ingestion, the defects in insulin-mediated glucose uptake by muscle and the suppression of glucose production by insulin contribute approximately equally to the disturbance in whole-body glucose homeostasis in T2DM. However, under euglycemic hyperinsulinemic conditions, EPG is largely suppressed and impaired muscle glucose uptake is primarily responsible for the insulin resistance.
You May Like: Getting Life Insurance With Type 2 Diabetes
X Assessing For Diabetes In The Patient With Nafld
Given the high prevalence of diabetes in patients with NAFLD, it is mandatory to screen patients with NAFLD for hyperglycemia. An early diagnosis of diabetes in patients with NAFLD would provide an opportunity to direct therapy toward insulin resistance and optimal glycemic control to create a favorable metabolic memory that would reduce the subsequent risk of diabetes complications specifically and also perhaps liver complications . Traditionally, screening has been done by formal laboratory-based fasting blood glucose levels. However, whereas the cost and resource demands involved in screening the large number of patients with NAFLD needs to be considered, the OGTT has been proposed as a better screening tool for diabetes in NAFLD. The OGTT improves the sensitivity of diabetes detection and also detects patients with IGT, a prediabetic state with established links to cardiovascular risk and early retinopathy .
Completely separate from the diagnosis of diabetes, the OGTT may also be useful for assessment of liver fibrosis risk by quantifying insulin and glucose levels after a fixed carbohydrate load . Thus, whereas insulin levels are not necessary nor are they used in the diagnosis of diabetes, as described in earlier sections, detecting hyperinsulinemia and insulin resistance parameters after a glucose load may help to assess risk of fibrosis and its progression in NAFLD in those with, and without, glucose dysregulation .
Vascular And Myocardial Insulin Resistance
The first and rate-limiting step in insulin-mediated glucose disposal is the transit of insulin from the plasma to the muscle. Crossing of insulin from the circulation into the muscle interstitium is governed by vascular endothelium. The transendothelial transport depends on the insulin receptor binding to the endothelial cell membrane and requires the activation of the nitric oxide synthase. The transport of insulin across the endothelial cell layer appears to involve a complex vesicular trafficking process, which is saturable. Insulin is known to promote capillary vasodilation particularly in the postprandial period to facilitate entry and distribution of fuel substrates, including glucose. Several studies sampling lymph and interstitial glucose, using dialysis techniques, have suggested that a delay in insulin transfer from the plasma to the tissue may play an important role in the development of insulin resistance . Thus, impairment of insulin action may be secondary to a decrease in capillary density or to a defective increase in blood flow or micro-capillary recruitment . These abnormalities have been described in obese insulin-resistant and in the skin flow response of patients with diabetes.
Don’t Miss: Can Diabetes Make You Lose Your Eyesight
T2dm Outcomes/complications: Cardiovascular Risk
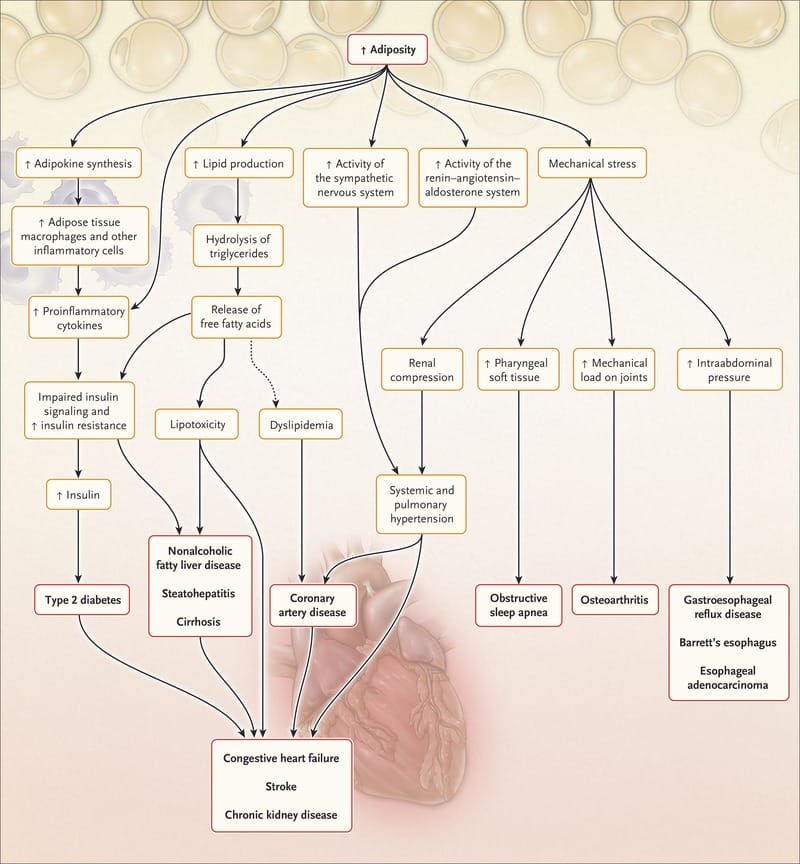
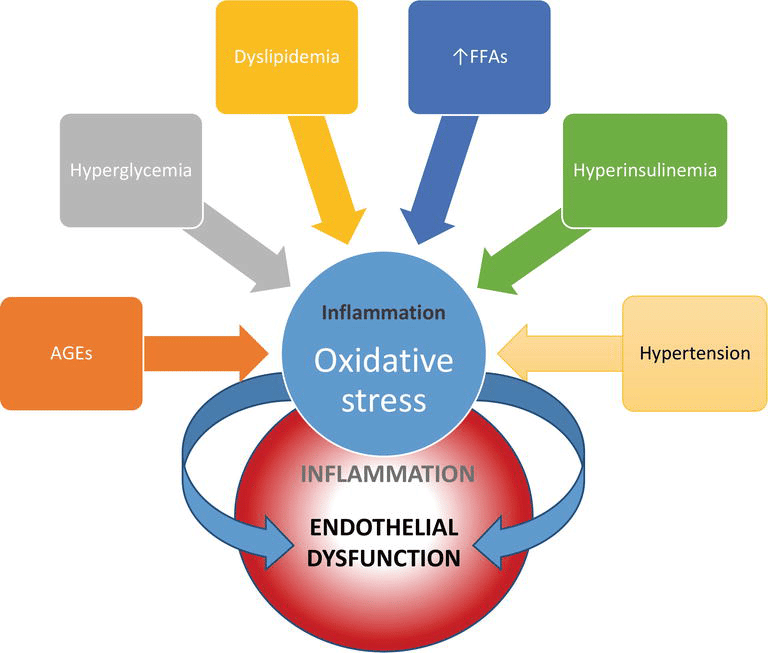
As described in the previous sections, T2DM is a multisystem disease with a strong correlation with CVD development . T2DM leads to a two- to four-fold increase in the mortality rate of adults from heart disease and stroke and is associated with both micro- and macro-vascular complications, the latter consisting of accelerated atherosclerosis leading to severe peripheral vascular disease, premature coronary artery disease and increased risk of cerebrovascular diseases . These factors lead to T2DM being considered a significant risk factor for CVD , likely through the involvement of several molecular mechanisms and pathological pathways. These include the role of IR in atherosclerosis, vascular function, oxidative stress, hypertension, macrophage accumulation and inflammation . The following sections describe in detail the main factors implicated in cardiovascular risk outcomes from T2DM and the interactions between them .
Factors implicated in cardiovascular risk outcomes from T2DM and the interactions between them. T2DM derived hyperglycemia, hyperinsulinemia and IR causes endothelial dysfunction, diabetic dyslipidemia and inflammation leading to CVD. The flowchart illustrates the multiple interactions among the implicated factors.
A Hepatic Insulin Resistance And Type 2 Diabetes
The liver, together with skeletal muscle and adipose tissue, displays exquisite sensitivity to insulin , with classical insulin signaling occurring mainly through specific cell surface receptors and series of postreceptor signaling pathways . By insulin-dependent processes, the liver dynamically regulates glucose flux and metabolism and, consequently, glycemia .
Hepatic insulin resistance is a common feature that predisposes to compensatory hyperinsulinemia, often followed by consequent pancreatic -cell dysfunction and development of type 2 diabetes . In recent years, the role of neural and endocrine signaling between the liver and brain in the pathophysiology of diabetes has also been more clearly defined .
Read Also: Is Mushroom Good For Diabetes
The Effect Of Stz Injection In The Transgenic Mouse Model Of Ad
The effects of DM as a result of systemic-STZ-induced insulin deficiency were evaluated in two transgenic models: pR5 mice with P301L tau and triple transgenic AD.29, 30 Aggravation of tau pathology by concomitant DM was evident, with markedly increased phosphorylation and insoluble fraction of tau forming neurofibrillary tangles.29 The same effect of DM on APP and soluble A was also noted, but not on the expression of total tau in the 3xTG AD model.30 Interestingly the amyloidogenic effect of DM was decreased by continuous supplementation of a glucagon-like peptide-1 agonist, one of the antidiabetic medications.
As described above, i.c.v. injection of STZ results in an insulin-resistant state in the brain.18, 19 Therefore, this model is useful for evaluating the effect of insulin resistance. Reduced cognition as well as increased cerebral aggregated A fragments, total tau proteins, and congophilic amyloid deposits were observed when Tg2576 mice injected with STZ at 3 months of age were evaluated at 9 months of age.31 Thus, both insulin deficiency and insulin resistance are thought to accelerate AD pathology.
C A Focus On Clinical Markers Of Nafld Progression Beyond Liver Biopsy Findings
Defining clinical risk factors and markers for NAFLD presence, severity, and particularly progression to significant fibrosis is of major clinical importance. This is required to aid the screening of subgroups at high risk for progression to significant liver disease, to increase our understanding of NAFLD and NASH pathogenesis, and to identify potential therapies for NAFLD, including the targeting of reversible pathogenic risk factors.
Don’t Miss: Can Drinking Water Lower Blood Sugar
A Nafld In Diabetes And Prediabetes
NAFLD prevalence is increased in people with type 2 diabetes and has been estimated at around 70% using ultrasound techniques . This figure is likely to be an underestimate because ultrasound has reduced sensitivity to detect NAFLD presence at levels of liver steatosis below 33% and also in more obese patients . Data on NAFLD in type 1 diabetes are far less comprehensive. A study by Targher et al. of 250 patients with type 1 diabetes estimated the prevalence of NAFLD by ultrasound at 44.4%.
Although studies in patients with type 2 diabetes examining the spectrum of pathology within NAFLD utilizing liver biopsy are limited by small numbers, they collectively estimate the prevalence of NASH at 6387% and moderate-severe fibrosis at 2260% . In a recent study of patients with type 2 diabetes, with an average BMI of 36 kg/m2, over 60% of patients who underwent weight reduction surgery had moderate to severe NAFLD on liver biopsy .
Diabetes presence is particularly associated with fibrosis in NASH . Angulo et al. found that when diabetes and obesity coexisted, 66% of patients with NAFLD had advanced fibrosis. This figure is much higher than those estimated for patients without diabetes or obesity . Furthermore, a study by Leite et al. in patients with type 2 diabetes found that three of the 92 patients had histological evidence of cirrhosis secondary to NAFLD without clinical evidence of liver disease.
| NAFLD pathological subcategory . |
|---|