Coverage And Correct Coding Of Continuous Glucose Monitoring Devices
Christine Woolstenhulme, QCC, QMCS, CPC, CMRSDecember 4th, 2014
Joint DME MAC Publication
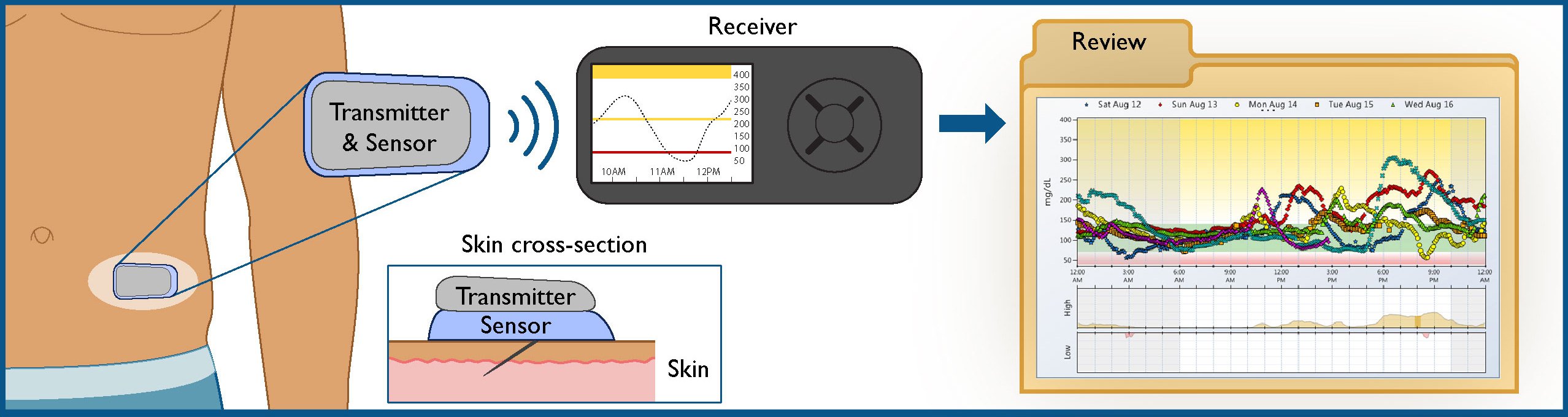
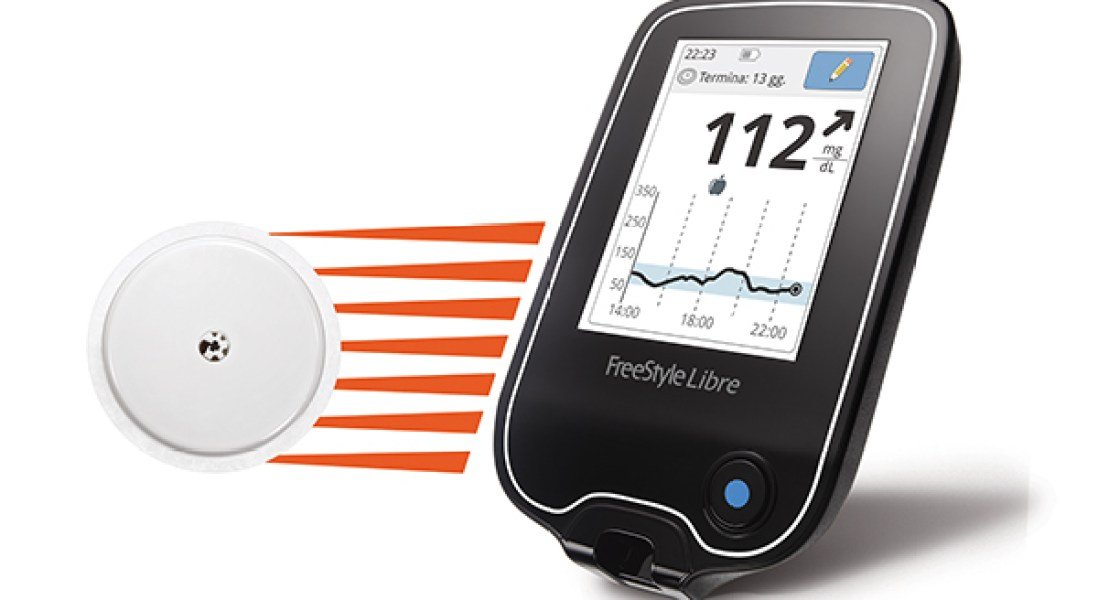
Continuous glucose monitoring devices measure glucose in the interstitial fluid, not capillary blood, providing interstitial glucose readings every few minutes. CGM systems are composed of several components disposable sensors that are inserted in the subcutaneous tissue, a transmitter that relays information to the receiver, and a receiver where the information is displayed.
COVERAGE
Current CGM systems are FDA-approved only as a secondary source for glucose monitoring. According to the FDA labeled indications, all CGM device readings must be confirmed with a capillary blood glucose monitor and users are cautioned against making insulin dosage changes based solely on CGM system determinations. Consequently, CGM devices are considered precautionary equipment. The Medicare Durable Medical Equipment Benefit excludes precautionary items from coverage therefore, claims for CGM systems are denied as statutorily non-covered, no benefit.
Medicare covers necessary supplies used with covered items. When the base item is non-covered, the related supplies are also not covered. Claims for supplies used with CGM systems are denied as statutorily non-covered, no benefit.
CODING
CGM systems are provided either as stand-alone systems or integrated into an insulin pump. For stand-alone systems and related supplies, use the following HCPCS codes:
E0784 – EXTERNAL AMBULATORY INFUSION PUMP, INSULIN
Hcpcs S1031continuous Noninvasive Glucose Monitoring Device Rental Including Sensor Sensor Replacement And Download To Monitor
1Two-digit numeric codes are Level I code modifiers copyrighted© by the American Medical Association’s Current Procedural Terminology .
2 BETOS stands for Berenson-Eggers Type Of Service
A modifier provides the means by which the reporting physician or provider can indicate that a service or procedure that has been performed has been altered by some specific circumstance but not changed in its definition or code.Modifiers may be used to indicate to the recipient of a report that:
- A service or procedure has both a professional and technical component.
- A service or procedure was performed by more than one physician and/or in more than one location.
- A service or procedure has been increased or reduced.
- Only part of a service was performed.
- An adjunctive service was performed.
- A bilateral procedure was performed.
- A service or procedure was provided more than once.
- Unusual events occurred.
Code used to identify the appropriate methodology for developing unique pricing amounts under part B. A procedure may have one to four pricing codes.
Code used to identify instances where a procedure could be priced under multiple methodologies.
A code denoting Medicare coverage status.
The Berenson-Eggers Type of Service for the procedure code based on generally agreed upon clinically meaningful groupings of procedures and services.
A code denoting the change made to a procedure or modifier code within the HCPCS system.
Effective date of action to a procedure or modifier code
New Cgm Codes And Billing Guidelines Effective April 1 2022
The Centers for Medicare & Medicaid Services issued a final rule on durable medical equipment that took effect on February 28, 2022:
- The final rule classifies non-implantable CGMs as DME, regardless of whether the CGM has been approved or cleared by the Food and Drug Administration to replace a blood glucose monitor for use in making diabetes treatment decisions.
- Adjunctive CGMs Adjunctive CGMs that do not replace a blood glucose monitor are referred to as adjunctive CGMs because they can be used as an adjunct to the blood glucose monitor by showing trends in glucose levels and alerting the patient about potentially dangerous levels, even while they sleep, that then must be verified by use of a separate blood glucose monitor.
- Non-adjunctive CGMs Non-adjunctive CGM receivers are DME that displays and monitors the continuous glucose readings and trends, and replace the use of a blood glucose monitor in diabetes treatment decision-making.
Also Check: What Type 2 Diabetes Medication Has The Least Side Effects
Personal Vs Professional Cgm
CGM devices are made of three parts: a sensor to detect interstitial glucose that is inserted subcutaneously by the patient or health care professional, a receiver or reader that displays the current glucose level, and a transmitter that sends data from the sensor to the receiver. CGM devices fall into two categories: personal use and professional use. Patients may purchase their own personal use CGM devices if they have Type 1 diabetes, uncontrolled Type 2 diabetes on intensive insulin regimens, or erratic hypoglycemia episodes. The devices help patients learn to respond to glycemic fluctuations and increase confidence in blood sugar management. They also allow patients to share their data remotely with family members, caregivers, or clinicians.
An in-depth review of personal CGM patient selection and device comparison has been published elsewhere.5 No long-term studies have been performed to determine whether personal use CGM improves clinical outcomes in patients with Type 2 diabetes however, evidence suggests both personal and professional CGM improve glycemic control by increasing time in range.6,7
Professional CGM can be beneficial for patients who aren’t interested in personal CGM , or it may serve as a trial run for patients considering a personal CGM device.
Ama Disclaimer Of Warranties And Liabilities
CPT is provided “as is” without warranty of any kind, either expressed or implied, including but not limited to, the implied warranties of merchantability and fitness for a particular purpose. AMA warrants that due to the nature of CPT, it does not manipulate or process dates, therefore there is no Year 2000 issue with CPT. AMA disclaims responsibility for any errors in CPT that may arise as a result of CPT being used in conjunction with any software and/or hardware system that is not Year 2000 compliant. No fee schedules, basic unit, relative values or related listings are included in CPT. The AMA does not directly or indirectly practice medicine or dispense medical services. The responsibility for the content of this file/product is with CGS or the CMS and no endorsement by the AMA is intended or implied. The AMA disclaims responsibility for any consequences or liability attributable to or related to any use, non-use, or interpretation of information contained or not contained in this file/product. This Agreement will terminate upon notice if you violate its terms. The AMA is a third party beneficiary to this Agreement.
Also Check: Diabetic Coma Is A Condition That Occurs When
Continuous Glucose Monitoring Systems: Leveraging Everyday Tech To Enhance Diabetes Management
However, is the coding for the treatment and management of diabetes being adequately captured?
Diabetes mellitus affects over 400 million people worldwide. It is a chronic disease of inadequate control of blood levels of glucose that affects the bodys ability to turn food into energy.
Essentially, the food you eat is broken down into sugar and released into the blood to be used as the bodys primary energy source. This increase in blood sugar causes the pancreas to release insulin, a hormone that acts like a key to enable the glucose to enter the bodys cells so it can be leveraged.
Lack of insulin or the inability of glucose to enter cells causes sugar to build up in the blood, which, over time, can lead to complications.
Diabetes has many subclassifications, but the two main types of DM are type 1 and type 2. Type 1 diabetes is typically diagnosed in children or younger adults, though it can develop at any age. With type 2 diabetes, patients problems begin when the cells in their body start to not respond to insulin as well as they should. This is called insulin resistance, which causes high blood sugar levels . This is the most common type of diabetes.
As noted, people with diabetes have trouble regulating glucose. Regularly checking blood glucose levels is important. Maintaining those levels within a target range helps improve energy and mood while preventing or delaying severe health complications.
Point And Click License For Use Of Current Dental Terminology
End User License Agreement
THE LICENSE GRANTED HEREIN IS EXPRESSLY CONDITIONED UPON YOUR ACCEPTANCE OF ALL TERMS AND CONDITIONS CONTAINED IN THIS AGREEMENT. BY CLICKING BELOW ON THE BUTTON LABELED “I ACCEPT”, YOU HEREBY ACKNOWLEDGE THAT YOU HAVE READ, UNDERSTOOD AND AGREED TO ALL TERMS AND CONDITIONS SET FORTH IN THIS AGREEMENT.
IF YOU DO NOT AGREE WITH ALL TERMS AND CONDITIONS SET FORTH HEREIN, CLICK BELOW ON THE BUTTON LABELED “I DO NOT ACCEPT” AND EXIT FROM THIS COMPUTER SCREEN.
IF YOU ARE ACTING ON BEHALF OF AN ORGANIZATION, YOU REPRESENT THAT YOU ARE AUTHORIZED TO ACT ON BEHALF OF SUCH ORGANIZATION AND THAT YOUR ACCEPTANCE OF THE TERMS OF THIS AGREEMENT CREATES A LEGALLY ENFORCEABLE OBLIGATION OF THE ORGANIZATION. AS USED HEREIN, “YOU” AND “YOUR” REFER TO YOU AND ANY ORGANIZATION ON BEHALF OF WHICH YOU ARE ACTING.
Read Also: Vitamin D And Type 2 Diabetes
Billing Information For Continuous Glucose Monitoring For Cpps
Effective Feb. 1, 2022, Clinical Pharmacist Practitioner taxonomy code 1835P0018X will be allowed to bill and be reimbursed for:
CPT code 95249 – Ambulatory continuous glucose monitoring of interstitial tissue fluid via a subcutaneous sensor for a minimum of 72 hours patient-provided equipment, sensor placement, hook-up, calibration of monitor, patient training and printout of recording. Reimbursement rate at $44.80.
CPT code 95250 – Ambulatory CGM of interstitial tissue fluid via a subcutaneous sensor for a minimum of 72 hours physician or other qualified health care professional provided equipment, sensor placement, hook-up, calibration of monitor, patient training, removal of sensor and printout of recording. Reimbursement rate at $122.63.
Both codes are used for the technical component of CGM, and covers patient training, glucose sensor placement, monitor calibration, use of a transmitter, removal of sensor and downloading of data.
ICD-10 Diagnosis Codes must accompany the claim:
The CPP Fee schedule lists the reimbursement information for CPPs.
The New World Of Glycemic Measures
For many years, there has been a tremendous focus on A1C as the preferred, and sometimes only, measure of glycemia and diabetes management. However, as it represents an average glucose level over approximately three months, it does not directly provide insight into high glucose levels, low glucose levels, or variability in between, though each of these can be quite important. For example, two patients can have identical A1C values, but one may have little variability, with only modest and infrequent hyperglycemia and hypoglycemia, while a second may have high degrees of variation, frequently shifting between profound hyperglycemia and hypoglycemia, while achieving the same average glucose level as the first patient. This is where the added metrics available from CGM can be especially useful.
Assessing and documenting these few metrics in a systematic way can help you maximize the insights that CGM can provide and streamline your efforts. Simply summarizing them in a note can be very helpful in this regard. Below are some suggested smart text or macros for two popular electronic health record systems that can be used or adapted to other systems.
| Epic SmartPhrase |
|---|
Read Also: Diabetic Low Cholesterol Meal Plan
Adjunctive Cgm Supplies And Accessories
Effective for claims with dates of service on or after April 1, 2022, CMS is creating the following HCPCS code to represent supplies used with an adjunctive CGM device that operates in conjunction with an insulin pump:
A4238 SUPPLY ALLOWANCE FOR ADJUNCTIVE CONTINUOUS GLUCOSE MONITOR , INCLUDES ALL SUPPLIES AND ACCESSORIES, 1 MONTH SUPPLY = 1 UNIT OF SERVICE
Modifiers
Suppliers are reminded that the use of the CG, KF and KX modifiers are required, as appropriate, with the HCPCS codes describing both adjunctive and non-adjunctive CGM devices and the associated supply allowance codes.
Correct coding is an essential element for correct claim payment. The Pricing, Data Analysis and Coding contractor maintains a variety of resources to assist suppliers in determining the appropriate code for Medicare billing. For questions about correct coding, contact the PDAC HCPCS Helpline at 735-1326 during the hours of 9:30 am to 5:00 pm ET, Monday through Friday. You may also visit the PDAC website to chat with a representative, or select the Contact Us button at the top of the PDAC website for email, FAX, or postal mail information.
User License Agreement And Consent To Monitoring
End User Agreements for Providers
Some of the Provider information contained on the Noridian Medicare web site is copyrighted by the American Medical Association, the American Dental Association, and/or the American Hospital Association. This includes items such as CPT codes, CDT codes, ICD-10 and other UB-04 codes.
Before you can enter the Noridian Medicare site, please read and accept an agreement to abide by the copyright rules regarding the information you find within this site. If you choose not to accept the agreement, you will return to the Noridian Medicare home page.
THE LICENSES GRANTED HEREIN ARE EXPRESSLY CONDITIONED UPON YOUR ACCEPTANCE OF ALL TERMS AND CONDITIONS CONTAINED IN THESE AGREEMENTS. BY CLICKING ABOVE ON THE LINK LABELED “I Accept”, YOU HEREBY ACKNOWLEDGE THAT YOU HAVE READ, UNDERSTOOD AND AGREED TO ALL TERMS AND CONDITIONS SET FORTH IN THESE AGREEMENTS.
IF YOU DO NOT AGREE WITH ALL TERMS AND CONDITIONS SET FORTH HEREIN, CLICK ABOVE ON THE LINK LABELED “I Do Not Accept” AND EXIT FROM THIS COMPUTER SCREEN.
IF YOU ARE ACTING ON BEHALF OF AN ORGANIZATION, YOU REPRESENT THAT YOU ARE AUTHORIZED TO ACT ON BEHALF OF SUCH ORGANIZATION AND THAT YOUR ACCEPTANCE OF THE TERMS OF THESE AGREEMENTS CREATES A LEGALLY ENFORCEABLE OBLIGATION OF THE ORGANIZATION. AS USED HEREIN, “YOU” AND “YOUR” REFER TO YOU AND ANY ORGANIZATION ON BEHALF OF WHICH YOU ARE ACTING.
LICENSE FOR USE OF “PHYSICIANS’ CURRENT PROCEDURAL TERMINOLOGY”, FOURTH EDITION
Consent to Monitoring
Recommended Reading: Sex And Diabetes Type 2
Setting Up Cgm In Your Practice
Here’s a step-by-step process for adding any of these devices to your practice.
Get administrative buy-in. Administrative support is one of the key elements for successfully implementing a clinic-owned CGM program. Administrators will need to understand the challenges of treating diabetes and the benefits of CGM. Mapping out the billing and reimbursement process, staffing needs, and projected use before purchasing CGM systems can make administrators more likely to buy in.
Designate a staff champion. Another key to a successful CGM program is identifying a staff champion within the clinic. This role can be filled by a medical assistant, licensed vocational nurse, dietitian, or diabetes educator. The main qualifications are enthusiasm for new projects and good patient education skills. The staff champion will be trained to select patients for the CGM program , use and maintain devices, insert sensors and provide instructions to patients, download and disseminate the results, and bill and code for services. Device manufacturers can provide training videos and educational materials for staff. Training a backup staff member may be helpful for when the champion is unavailable.
| Diagnosis | |
|---|---|
| Labile blood glucose | R73.09 |
Billing For Continuous Glucose Monitor
On December 28, 2021, the Centers for Medicare & Medicaid Services published a final rule which was implemented on February 28, 2022. This final rule addressed the classification and payment of continuous glucose monitor under the Medicare Part B benefit for durable medical equipment . This rule expanded the classification of DME to a larger group of CGMs, regardless of whether the CGMs are non-adjunctive or adjunctive . As such, claims for adjunctive CGMs and related supplies and accessories can now be covered under the Part B DME benefit category when the system meets the DME definition.
You May Like: What Kind Of Syringes Do Diabetics Use
Trends In Coverage For Continuous Glucose Monitoring
In the case of an emerging category of technology, there is an educational period when payers must be properly convinced of the inherent value in providing the new technology to eligible patients. As continuous glucose monitoring continues to evolve, critical factors including clinical efficacy, improvement in net health outcomes, usability, and sustainability will play an important role in how payers respond to this evolving category of technology.
In the early 2000s, several leading commercial payers provided an initial favorable response to some of the earlier CGM technologies. For example, most commercial plans provided coverage for retrospective, intermittent use of continuous glucose monitoring systems purchased by the physician. In 2007, Humana Inc., Cigna Corp., and Aetna Inc. all reaffirmed their positive decisions for retrospective CGMS in policy updates for the CGM category, but stopped short of expanding coverage to include patient-use CGM technologies in the home setting beyond a one-sensor life cycle.
The last three factors emphasize the critical importance of patients and health care professionals in determining the future success of the CGM category. Improving the reimbursement environment for continuous glucose monitoring will be directly related to whether or not patients are properly educated and trained on how to use these sophisticated systems accurately and efficiently in order to optimize CGM as an effective tool in overall diabetes management.
Other Billing Details For Cgm
- Document the primary diagnosis code and the appropriate ICD indicator based on the date of service. Example diagnosis code: 250.03 (Diabetes without mention of complications type 1, uncontrolled. Common ICD-10 codes include:
- E10.649 Type 1 diabetes mellitus with hypoglycemia without coma
- E10.65 Type 1 diabetes mellitus with hyperglycemia
- E10.69 Type 1 diabetes mellitus with other specified complications
- E10.8 Type 1 diabetes mellitus with unspecified complications
- E10.9 Type 1 diabetes mellitus without complications
- E11.649 Type 2 diabetes mellitus with hypoglycemia without coma
- E11.65 Type 2 diabetes mellitus with hyperglycemia
- E11.69 Type 2 diabetes mellitus with other specified complications
- E11.8 Type 2 diabetes mellitus with unspecified complications
- E11.9 Type 2 diabetes mellitus without complications
- E13.8 Other diabetes mellitus with unspecified complications
- E13.9 Other specified diabetes mellitus without complications
Don’t Miss: Doterra Essential Oils For Diabetes